Gel Mobility Shift Assays Can Be Used to Determine
Electrophoretic mobility shift analysis, sometimes referred to as EMSA, is a sensitive method for investigating nucleic acid-poly peptide bounden. This is based on gel electrophoresis and is based on the departure in the way nucleic acid-protein complexes and costless nucleic acids move during gel electrophoresis or the difference in electrophoretic mobility.
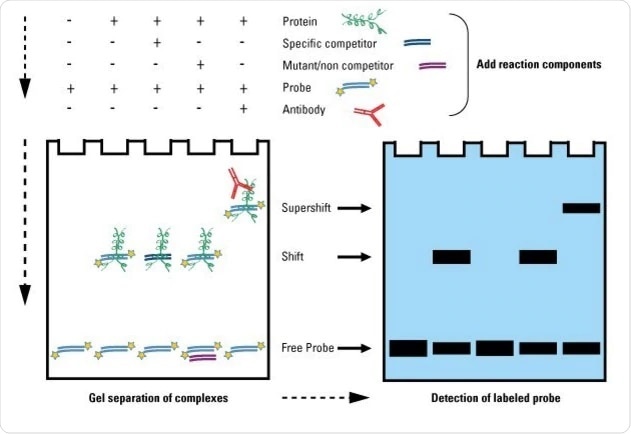
Paradigm Credit: https://world wide web.thermofisher.com/united kingdom/en/domicile/life-science/poly peptide-biology/poly peptide-biology-learning-eye/poly peptide-biology-resource-library/pierce-protein-methods/gel-shift-assays-emsa.html
There are many protocols for electrophoretic mobility shift assays, with ane involving using two-color fluorescence being used to visualize not only the nucleic acids merely too the protein.
How does electrophoretic mobility shift assay work?
This is an example of an electrophoretic mobility shift analysis using radioactively labeled DNA.
A 32P-labeled fragment of Deoxyribonucleic acid containing the potential protein binding site is mixed with the protein of interest in a buffer arrangement, consisting of HEPES-KOH, glycerol, EDTA, KCl, phenylmethylsulfonyl fluoride, and DTT. In one case incubated, this is straight loaded into the gel.
Typically, a not-denaturing polyacrylamide gel is used and this needs to exist "pre-run" beforehand. The gel can at this stage be stock-still using a solution of methanol and acetic acrid, which is optional. The gel is and so stale earlier existence exposed to autoradiography motion picture.
The binding of the protein makes the nucleic acid-protein complex motility slower than free nucleic acids, thus one time the gel has visualized the difference in band location indicates whether the poly peptide of interest has spring to the potential target DNA.
It is not always necessary to use radioactively labeled nucleotides; fluorophores can be used, so that fluorescence becomes the method of visualization. Another instance is the apply of biotin, which tin can be detected past either chemiluminescence or chromophore deposition. Alternatively, fluorophores or chromophores tin be added later on the electrophoresis, pregnant that the target nucleic acid does non need to be labeled.
Jing and co. developed a fluorescence-based electrophoretic mobility shift assay that used fluorophores to not only visualize the nucleic acids but as well the protein as well. This was accomplished by using ii fluorophores, SYBR Greenish for the nucleic acids and SYPRO Ruby for the proteins, which results in green and ruby fluorescence.
To validate the analysis, the authors used a 40 nucleotides fragment from the lac operon operator alongside the lac repressor protein at varying concentrations. After incubation, these samples were then put into a 6% non-denaturing polyacrylamide gel. One time the electrophoresis was completed, the gel was stained with SYBR Dark-green EMSA stain and visualized.
Following this, the gel was further stained with SYPRO Scarlet EMSA stain and visualized again. The images tin be overlaid together so that both fluorescent stains can exist visualized at the aforementioned fourth dimension.
The resulting gel showed that when the lac repressor binds to the target Deoxyribonucleic acid sequence in the lac operon operator, this results in yellow color in the overlay image. The intensity of these bands' changes with the concentration of the lac repressor protein, which is also seen with the SYPRO Reddish staining.
Therefore, this two-color fluorescence electrophoretic mobility shift assay is capable of detecting both nucleotides and protein in an electrophoretic mobility shift assay.
What are the advantages and limitations of electrophoretic mobility shift assays?
The advantages of electrophoretic mobility shift assay include simplicity, ability to be performed in a wide range of weather condition, and high sensitivity. This means that electrophoretic mobility shift assays are unproblematic to perform, where the analysis conditions can be modified within a wide range to conform may nucleic acid-poly peptide interactions; the temperature range is between 0˚C and sixty˚C, the pH range between iv and nine.5, and table salt concentrations can range from 1mM to 300nM to give some examples.
The high sensitivity means that the concentrations of the nucleic acids and proteins tin be lower than 0.1nM, and as information technology is an electrophoresis-based assay the sample book tin be less than 20μl.
While there are advantages, there are also limitations; these include the possibility of nucleotide-protein complex dissociation during electrophoresis, the possibility that the electrophoretic mobility of the nucleotide-protein complex is non just influenced by its size, and the inability of electrophoretic mobility shift assay to detect specific nucleotide sequences to which the protein binds.
What considerations are needed when performing an electrophoretic mobility shift assay?
While a standardized protocol may be sufficient for some electrophoretic mobility shift assays, some considerations need to exist taken
Selecting the target nucleic acid
Short fragments of DNA or RNA are cheap and quick to obtain and make it less likely for non-specific binding to occur. This is peculiarly advantageous if the protein of interest has a low specificity towards its target nucleic acid.
However, care needs to be taken to ensure that binding sites are not too close to the ends, as this can lead to structural and electrostatic end effects, ultimately resulting in abnormal binding during electrophoretic mobility shift assays.
Competing proteins
Nucleic acid sequences can take more than one protein that binds to information technology, which is often the case. This may not be a problem, but sometimes the reaction of involvement can be masked by the activity of a secondary poly peptide. In this case, unlabeled nucleic acids can exist added to compete with the labeled nucleotides. For this to be successful, the secondary protein needs to bind both nucleotides equally well, while the target poly peptide needs to have a higher affinity for the labeled nucleotides.
Bounden conditions
Salt concentrations can impact protein-nucleic acid bounden; therefore, this needs to be adjusted for the specific nucleic acid-protein complex before the electrophoretic mobility shift assay for the assay to be optimal.
The ability for electrophoretic mobility shift assays to be performed at a wide range of common salt concentration should enable many nucleic acrid-protein complexes to be analyzed past electrophoretic mobility shift assays.
Sources:
- (2005) Electrophoretic mobility shift assays Nature Methods https://doi.org/ten.1038/nmeth0705-557
- Hellman, L. One thousand., and Fried, Thou. G. (2007) Electrophoretic Mobility Shift Assay (EMSA) for Detecting Protein-Nucleic Acid Interactions Nat Protoc doi: ten.1038/nprot.2007.249
- Jing, D., et al. (2004) The utility of a two-color fluorescence electrophoretic mobility shift assay procedure for the assay of DNA replication complexes Electrophoresis https://doi.org/ten.1002/elps.200405994
Source: https://www.news-medical.net/life-sciences/What-is-an-Electrophoretic-Mobility-Shift-Assay.aspx
0 Response to "Gel Mobility Shift Assays Can Be Used to Determine"
Post a Comment